Understand who qualifies for resmetirom, MASH treatment eligibility, and fatty liver medication criteria
If you or your patients have been diagnosed with fatty liver disease that has progressed to MASH, one of the most common questions is:
Who qualifies for resmetirom?
Resmetirom, sold under the brand name Rezdiffra, is the first FDA-approved medication for MASH (formerly NASH) and has generated enormous interest among patients looking for an effective fatty liver treatment. (Medscape)
However, not everyone with fatty liver disease is eligible.
In this guide, we’ll explain exactly who should take resmetirom, the official eligibility criteria, and which patients are most likely to benefit.
This guide will cover the following queries:
- Who Qualifies for Resmetirom? Complete Eligibility Guide for MASH Patients
- Resmetirom Eligibility Criteria Explained: Who Should Take It?
- Fatty Liver Medication Criteria: Who Is Eligible for Rezdiffra?
What Is Resmetirom?
Resmetirom is an oral once-daily medication approved for adults with:
- MASH (Metabolic dysfunction-associated steatohepatitis)
- moderate to advanced liver fibrosis
- non-cirrhotic liver disease
It works by activating thyroid hormone receptor-beta (THR-β) in the liver, helping reduce:
- liver fat
- inflammation
- fibrosis progression (PMC)
This is a major advancement because until recently, diet, exercise, and weight loss were the only standard treatments.
Who Qualifies for Resmetirom?
This is the most important section for SEO and user intent.
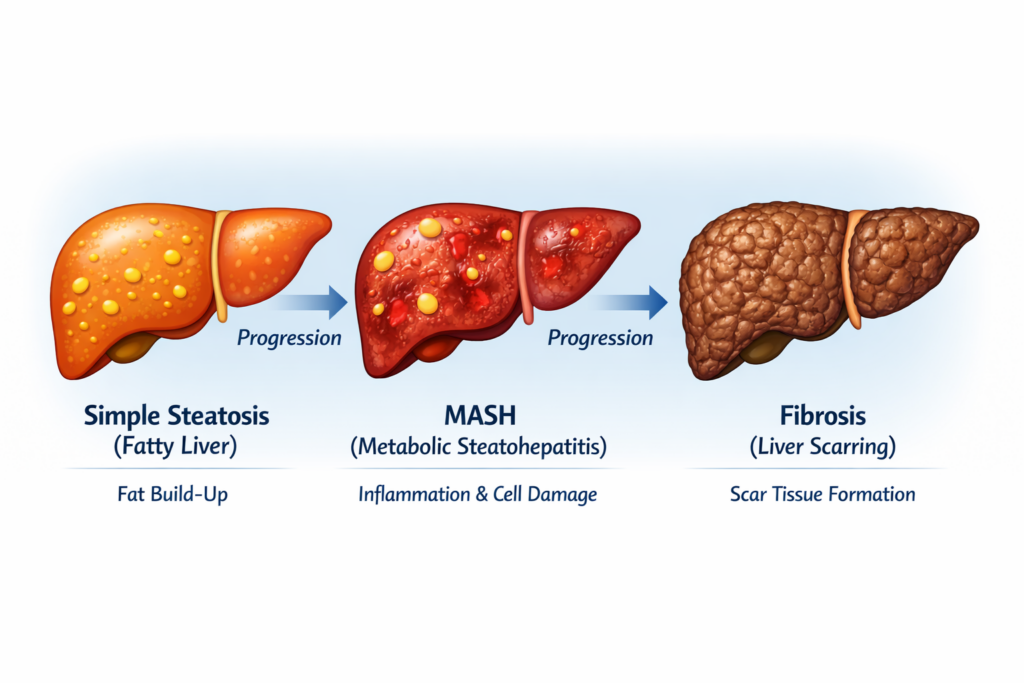
1. Adults Diagnosed With MASH / NASH
The first eligibility requirement is a confirmed diagnosis of:
- MASH
- previously called NASH (Nonalcoholic steatohepatitis)
This means the patient must have:
- fat accumulation in the liver
- liver inflammation
- liver cell injury
Simple fatty liver alone is not enough.
People with only fatty liver (MASLD / NAFLD) but without inflammation or fibrosis usually do not qualify. (PMC)
This is one of the biggest misunderstandings online.
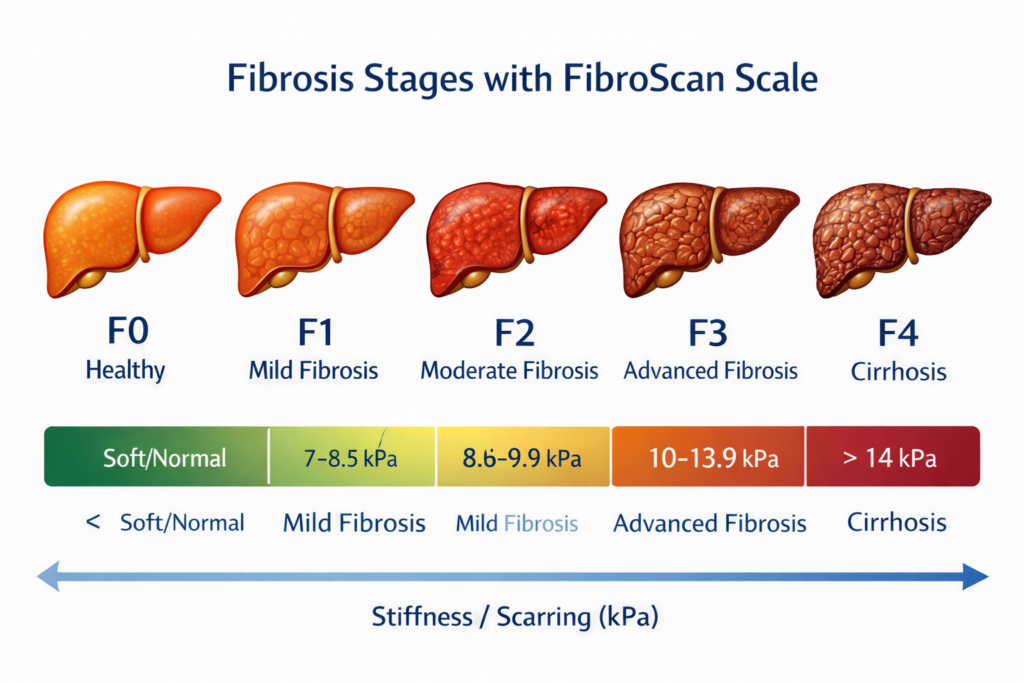
2. Fibrosis Stage F2 or F3
This is the main eligibility criterion.
Patients should have:
- F2 fibrosis (moderate scarring)
- F3 fibrosis (advanced scarring)
Resmetirom is specifically approved for:
adults with non-cirrhotic MASH with moderate to advanced fibrosis (F2–F3) (Medscape)
What This Means
Fibrosis stages include:
- F0: no scarring
- F1: mild scarring
- F2: moderate scarring
- F3: advanced scarring
- F4: cirrhosis
Patients in F2 and F3 stages are considered ideal candidates.
This group has the highest risk of progression to cirrhosis.
3. No Cirrhosis (Not for F4 Patients)
This is extremely important.
Resmetirom is approved for non-cirrhotic MASH only.
That means patients with:
- F4 fibrosis
- established cirrhosis
- liver failure signs
may not be eligible.
The drug should be avoided in decompensated cirrhosis. (PMC)
This includes patients with:
- ascites
- jaundice
- variceal bleeding
- hepatic encephalopathy
These patients need specialist hepatology care.
4. Evidence From FibroScan, MRE, or Liver Biopsy
Many users ask:
“Do I need biopsy for resmetirom?”
The answer is: not always.
Current guidance allows eligibility assessment through non-invasive liver tests, including:
- FibroScan (VCTE)
- MRI elastography
- liver fibrosis blood panels
- liver biopsy in selected cases (mash.pocn.com)
This is excellent for search traffic because many patients are actively searching these terms.
Common Tests Used
FibroScan
Measures liver stiffness.
MRI Elastography
More advanced imaging test.
Liver Biopsy
Gold standard, but not always required.
5. Patients With Metabolic Risk Factors
Most people eligible for resmetirom also have metabolic risk factors such as:
- obesity
- insulin resistance
- type 2 diabetes
- high triglycerides
- metabolic syndrome
- hypertension
These conditions strongly increase MASH risk.
Patients with central obesity and diabetes need to know this on priority.
Who Should NOT Take Resmetirom?
This section helps you understand who can and who cannot take the medicine.
Patients With Simple Fatty Liver Only
If the liver only shows fat but no inflammation/scarring, the medication is usually not indicated yet. (Reddit)
Patients With Cirrhosis
Advanced cirrhosis patients need specialist management.
Pregnant Women / Special Cases
Patients should consult their physician regarding:
- pregnancy
- thyroid disorders
- severe liver dysfunction
- drug interactions
Can Lifestyle Changes Replace Resmetirom?
The answer is: sometimes yes, sometimes no.
Lifestyle interventions remain essential:
- weight loss
- exercise
- Mediterranean diet
- intermittent fasting
- insulin resistance management
Even when taking resmetirom, it must be used along with diet and exercise. (Medscape)
Best Candidates for Resmetirom
The ideal candidate is someone who has:
- confirmed MASH
- fibrosis stage F2 or F3
- obesity or diabetes
- elevated liver enzymes
- high risk of progression
Have you got satisfactory answer for your query of: “who qualifies for resmetirom”
Final Thoughts
Resmetirom is a major breakthrough for patients with progressive fatty liver disease.
But eligibility is specific.
The best candidates are adults with:
- MASH
- F2–F3 fibrosis
- no cirrhosis
- metabolic risk factors
If you are unsure whether you qualify, ask your hepatologist about:
- FibroScan
- fibrosis stage
- MASH confirmation
- treatment suitability
Early treatment may help prevent progression to cirrhosis.
After 38 Years of Medical Practice I have started to share insights from my experience over this blog. Know more about me here on About Page and Visit my LinkedIn profile.
